News
Archive for the ‘News’ Category
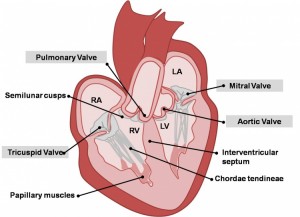
What is a Heart Murmur?
by admin on March 10th, 2021
Category: News, Tags:
- Murmurs explained
- FAQs about murmurs
- List of the common causes of murmurs in dogs and cats
- Murmurs explained
- FAQs about murmurs
- List of the common causes of murmurs in dogs and cats
- Mitral Valve Disease (MVD) is the most common cause of a murmur in adult dogs in older age
- Mitral Valve Dysplasia is a congenital defect of the valve
- Mitral Valve Regurgitation can occur secondary to heart enlargement of other causes in dogs
- Mitral Valve Regurgitation can be secondary to cardiomyopathy in cats
- Subaortic Stenosis and Valvular Aortic Stenosis is a congenital defect of the valve
- Hypertrophic Cardiomyopathy in cats, often causes ‘Subaortic- like Stenosis’
- Tricuspid Valve Disease occurs in adult dogs in older age, most will also have MVD
- Tricuspid Valve Dysplasia is a congenital defect of the valve
- Tricuspid Valve Regurgitation can occur secondary to Pulmonary Hypertension
- Tricuspid Valve Regurgitation can be secondary to heart enlargement of other causes
- Tricuspid Valve Regurgitation, when mild, can be seen in some normal dogs
- Valvular Pulmonic Stenosis is a congenital defect of the valve
Murmurs associated with holes in the heart
- Patent Ductus Arteriosus is a congenital defect resulting in abnormal flow between the aorta and pulmonary artery
- Ventricular Septal Defect is a congenital defect in which there is a hole between the two ventricles
- Puppy murmurs are innocent murmurs that usually disappear by 4-6 months of age
- Anaemic murmurs are due to the blood being thin
- Fever murmurs are due to turbulence in blood flow
- Flow murmurs are seen in some athletically fit dogs, ie. a murmur with no defect present
COVID-19 UPDATES
by admin on July 7th, 2020
Category: News, Tags:
We are pleased to report that our Littlehampton Clinic is open and we have resumed services with social distancing measures in place.
We are allowing our two legged friends into the practice with their pets but our passageways and consulting rooms are narrow so in order to maintain social distancing we ask that you notify us when you have arrived, by phone or tapping on the window, and remain outside until invited to enter, to avoid crowding.
Hand hygiene stations have been installed on entry to the main building and at reception, as well as screens at reception.
Please respect our social distancing measures which will include asking that only one owner accompanies per pet, and highlight that our preferred payment method is by card. We request face coverings and you may be asked to wait outside if you do not have one.
In the consulting room we will ask you to remain seated whilst a vet and nurse examine your pet.
We request that wherever possible repeat medications are paid for in advance over the phone by card, so that interaction is kept minimal on collection.
Due to the ways of working we have had to adopt to keep our staff, you and your pets safe, things are taking us a bit longer, reducing the number of pets we can see in practice every day. Please bear with us while we continue to prioritise in-practice appointments and operations, and offer video or telephone consults for some pets.
We hope to re-open our branches in due course. Social distancing requirements mean that for the time being our clients may not hold their pets during examination. We are therefore having to recruit a nurse to assist in this capacity and will probably need to introduce appointments to avoid crowding when we re-open in Arundel and Angmering.
We would like to say a massive THANK YOU to all our customers for being so understanding and accommodating at this difficult time.
Covid-19 Update & Out of Hours Provision
by admin on June 16th, 2020
Category: News, Tags:
Transitioning to a more normal range of veterinary services:
In line with the easing of restrictions across the UK, our governing body are allowing us to begin transitioning towards providing as full a range of veterinary services as possible in a safe manner. However, during the pandemic it will not be a return to pre-Covid ‘business as usual’. We will be increasing our services this week by degrees, and in relation to demand.
Safe working:
All our services have to be risk assessed and provided in a manner that supports safe working and recognises 2m Social Distancing. Our premises do not lend themselves to the necessary social spacing that is possible in supermarkets and high-street stores, nor do our consulting rooms.
Visiting the clinic:
Creating a socially-distanced queueing system in our limited space would seriously delay pets arriving for urgent appointments so we respectfully request that all clients arriving at the clinic do not enter but instead:
- Call us, confirm their mobile phone number, and supply their car type and registration if applicable
- Clients without mobile phones, please knock on the window
- Please be prepared to remain outside the clinic e.g. in a car
Pets will be collected from holding zones and examined. The vet will then contact you and treatment plans will be agreed remotely. Payment can be made over the phone.
Contact-free collection of medication, pet food and anti-parasitic medications is in place and a specific, secure collection time and place can be organised in advance. For any products you’d like to order simply call us to arrange for payment and safe collection of these items.
Where appropriate you will be offered video or telephone consults, enabling you to get the advice you need and for our team to decide if we need to see your pet in person to provide further care. For any emergencies or seriously ill pets we will ask you to attend the practice.
Your safety and the safety of our teams is our top priority so please follow the social distancing procedures we have in place to minimise contact.
We apologise that formerly simple procedures and transactions are taking much longer than was usual, for reasons of social distancing. Your co-operation and patience are greatly appreciated.
Out of Hours Services:
From Monday 15th June our out of hours emergencies will be attended by Worthing Emergency Vets based at Grove Lodge Veterinary Hospital in Worthing, West Sussex, for a trial period of 3 months. Please rest assured that all pets in our hospital will continue to be attended by one of our vets and nurses outside opening hours, and will not be transferred to Worthing.
At the end of March two of our team of vets departed for new positions but were not replaced because of lockdown. The remaining small team of vets continued to provide emergency cover. We are sad to say that this reduced team are no longer able to continue to provide 24 cover whilst at the same time returning to normal working hours, and the volume of work the clinic is currently allowed to undertake does not allow for replacement of these team members at present.
COVID-19 Update & article on Feline Lower Urinary Tract Disease
by admin on May 5th, 2020
Category: News, Tags:
Our main Littlehampton hospital REMAINS OPEN and in line with government guidelines small teams are working 24-hour shifts to provide emergency care and urgent treatment where animal welfare would be compromised by delay, as well as providing repeat medicines.
Concerned about your pet’s health but don’t think it’s an emergency?
If you are worried about your pet, please phone us and explain your situation. We’ll arrange a suitable way of conducting your appointment that is in everyone’s best interests.
There are a variety of options available to everyone – the important thing is to let us know and we’ll work together!
To respect social distancing rules, we are replacing our standard consultation service with video and telephone consultations. Please contact the practice to arrange an appointment and discuss suitable communication platforms (eg Skype, Facetime, WhatsApp). Payment can be made over the phone.
What constitutes an emergency that needs immediate care?
If any of the below apply to your pet, contact us IMMEDIATELY:
- Breathing difficulties
- Open wound injuries
- Trauma (e.g. car accidents)
- Male cats struggling in the litter tray to pass urine
- Rabbits neglecting food
- Swallowing hazards ie.toys/clothes
- Ingestion of poisonous/harmful substances
- Eye problems
- Vomiting or diarrhoea especially if your pet appears quiet or depressed or if it is going on for a long time (more than 24 hours)
- Swollen abdomen or retching (especially large dogs)
- Loss of thirst & appetite
- Struggling to give birth
- Seizuring/fitting
- Collapse
If the recommendation is to come into the practice, please note the following advice to keep you and our staff safe:
- We will ask that you telephone us when you arrive in the car park.
- A member of staff will come out and take your pet into the clinic.
- After making an assessment we will ring you on your mobile to discuss the necessary treatment and way forward.
Can I bring my pet in for an INITIAL VACCINATION COURSE?
Unvaccinated puppies, kittens and rabbits are at risk from picking up the diseases we commonly vaccinate against.
Whilst the risk can be lessened by keeping pets inside and avoiding contact with unvaccinated animals, please contact us to discuss your pet’s current situation with you, and risk assess whether vaccination at this point is recommended.
Can I bring my pet in for their ANNUAL VACCINATION BOOSTER?
We can postpone this vaccine for up to 3 months after the due date without requiring a restart. During these exceptional times, we ask that you delay the booster, but for no more than a maximum of 3 months after the due date.
Can I bring in my pet for a HEALTH CHECK?
Not at this moment in time because this is not deemed critical. However, if your pet is ill we will be able to conduct a consultation via a video or telephone consultation. Please ask the practice for further information.
Can I bring my pet in for NAIL CLIPS? Or to have their ANAL GLANDS expressed?
This should be postponed, provided your pet is not suffering pain or discomfort.
How can I get my REPEAT FOOD or MEDICATIONS?
It is important that you continue to give your pet the appropriate food and medication. We are looking at ways of getting these to clients who are due them and postal / delivery services may be an option. Please contact us to make suitable arrangements.
How can I get my FLEA, WORM, TICK and FLYSTRIKE treatments?
It is important that you continue to give your pet the appropriate treatments. We are looking at ways of getting these to clients who are due them and postal / delivery services may be an option. Please contact us to make suitable arrangements.
Can my pet still have ELECTIVE OPERATIONS such as Neutering and Lumps removals?
These operations are not deemed to be critical during these extraordinary times and we therefore ask that unless your pet’s health is being endangered, you postpone these procedures until government guidelines on social distancing change. If you have specific concerns please contact us to discuss your pet’s current situation, and risk assess whether surgery at this point is recommended.
What about ULTRASOUNDS and BLOOD TESTS?
It depends whether these are deemed critical or not – please contact us to discuss.
Please continue taking care of yourselves and all your lovely pets!!
WHAT IS FLUTD?
FLUTD stands for feline lower urinary tract disease. This is a term used to describe a collection of conditions which can affect the bladder and/or urethra (tube from the bladder to the outside).
The most common conditions causing FLUTD are:
- Urolithiasis (stones in the bladder and/or urethra)- there are various different stones which can form in the urinary tract resulting in FLUTD due to irritation of the bladder/urethral lining.
- Structural defects- rarely some cats can be born with anatomical defects which can lead to signs of FLUTD. Alternatively a defect such as a stricture (narrowing of the urethra secondary to scar formation) can develop in older animals.
- Bacterial infection- this is a relatively uncommon cause of FLUTD. In some cases an infection can be present alongside another condition.
- Cancer- tumours of the bladder and/or urethra are thankfully rare in cats.
- Urethral plugs and urethral spasm- urethral plugs are accumulations of cells, cyrstals and other debris which, if formed in the urethra, can block urine output from the bladder in male cats. In addition spasm (muscular contraction and narrowing) of the urethra can occur which can also result in blockage of the urethra.
- Feline Idiopathic Cystitis (termed F.I.C.)- this is the most common cause of FLUTD (around 60-70% of cases). The exact cause of F.I.C. is unknown although there are studies which suggest that this condition results from an abnormal response to stress. This condition is diagnosed after excluding all other causes however many cats with other conditions (for example urethral plugs/spasm) will also have F.I.C.
What are the signs of FLUTD?
The most common clinical signs of FLUTD relate to abnormalities of urination. In some cats which only urinate outdoors signs may not be apparent at home. If observed signs would include:
- Straining to urinate
- Crying when urinating
- Urinating in inappropriate places
- Urinating more often than normal
- Smaller volumes of urine produced when urinating
- Inability to urinate (this can be life threatening, requiring immediate veterinary attention).
- Blood visible in the urine
- Over-grooming the perineum
- Signs of pain/inappropriate aggression particularly around the hind end.
How is FLUTD investigated?
Initially a thorough history including identification of any potential factors that could lead to stress (i.e. house move, new pet in the environment or building work at home) is taken. A physical examination will then be performed. In male cats the bladder size and texture will carefully be assessed to identify whether urethral blockage (obstruction) is present. Next a combination of some or all of the following tests may be performed:
- Urinalysis: This may be collected at home (free-catch) or by taking a sample directly from the bladder using a needle. The urine concentration is assessed alongside checking for bacteria, blood, protein or crystals.
- Blood tests: A blood sample may be taken, especially in cats with suspected urethral blockage. This is to assess kidney function and blood salt (electrolyte) concentration. In addition some cats can have alterations in blood calcium levels which could predispose to stone formation.
- Abdominal ultrasound: This can be used to check for any signs of a tumour or stones in the bladder. The bladder wall thickness can also be assessed.
- X-rays: Some bladder or urethral stones (uroliths) can be visible on an X-ray of the abdomen.
How is FLUTD treated?
The treatment of FLUTD will vary depending on the underlying cause. In general FLUTD is a painful condition and therefore in all cases pain relief is normally administered. A short summary of possible treatments for each cause is given below.
Urolithiasis
If bladder stones are causing clinical signs they may need to be removed. Some types of stones (e.g. struvite) can be dissolved by changing to a specifically manufactured prescription diet. Others are normally removed surgically. If a urethral stone is present and causing blockage then additional, emergency treatment is required. This is discussed in more detail in the “Urethral plugs and urethral spasm” section.
Structural defects
Often surgery is required to correct structural defects. In cats with urethral strictures, dependent on the location, a procedure called perineal urethrostomy can be performed to open the urethra above the narrowing, creating a permanent new location for urine to exit.
Bacterial infection
Bacterial infections are treated with antibiotics. Due to the increasing prevalence of resistance, antibiotics should only be used in cats where there is either a very strong suspicion of infection or confirmed infection (particularly as this is less common).
Cancer
The most common type of cancer found in the bladder is called transitional cell carcinoma. This is generally treated with a combination of chemotherapy and non-steroidal anti-inflammatories. Other benign tumours may be treated by surgical removal.
Urethral plugs and urethral spasm
Urethral plugs or spasm resulting in blockage of the urethra (normally in male cats due to the diameter, shape and length of the urethra) can be life threatening. This is because if it is left untreated the kidneys can become damaged and blood salts can become deranged leading to heart problems and even death.
Cats with urethral blockage are rapidly assessed. Normally pain relief and muscle relaxants are administered alongside fluid therapy into the vein. The patient is then either anaesthetised or heavily sedated and removal of the blockage is performed (normally by flushing and placement of a catheter through the urethra into the bladder). After the catheter is passed into the bladder it is often secured in place for a few days. This is prevent immediate re-blockage and to allow time for the spasm and associated inflammation/swelling of the urethra to improve. The catheter is then removed and the patients are observed to ensure that they are able to urinate.
In cats with kidney problems due to the blockage fluid therapy into the vein will need to be continued until the kidneys improve. Sadly in some cases permanent damage to the kidneys can occur resulting in chronic kidney failure. Thankfully this is uncommon.
Rarely it is not possible to unblock the urethra with a catheter. In addition some cats have recurrent episodes of urethral blockage despite preventative therapy. In this specific but small group of patients a perineal urethrostomy can be performed (as discussed in structural defects). It is important to understand that there can be serious complications associated with the surgery and it does cure the condition.
Feline Idiopathic Cystitis (F.I.C.)
The management of F.I.C. is complex as the cause is multifactorial. Factors thought to contribute to the condition include a defective bladder lining, stress (specifically an individual’s inappropriate reaction to stress) and inflammation caused by the central nervous system. In humans a similar condition exists called ‘interstitial cystitis’. Changes in the bladder lining and brain of cats with F.I.C. are similar to those found in humans with this disease.
There are various different treatments available, with evidence behind each recommendation varying in quality and quantity. A short summary of each therapy area is given below:
- Dietary medication: Simply changing from dry to wet (sachet or tinned) food can help to increase the amount of water consumed. This can result in the production of more dilute urine. There are various prescription urinary diets available which may be recommended in specific circumstances (particularly if struvite stones are present or high blood calcium).
- Increasing water intake: In addition to changing to wet food, cats with F.I.C. should be encouraged to drink more water to enhance the production of dilute urine. This can be achieved by providing a water fountain, ensuring fresh water is always available in multiple locations or adding water to food.
- Litter tray number and location: There should be at least one litter tray for each cat in the household. These should be placed in different locations, preferably in less busy areas of the home. The type of litter can also have an impact and therefore providing a few different types to evaluate which is preferred can help. Cats that normally urinate outside should not be forced to use a litter tray.
- Identification and modification of stressful stimuli: This is a very important part of management but is often the most difficult to practically achieve. If an event that would be anticipated to cause stress is to occur (i.e. house move or new pet) then measures to minimise the impact should be considered. It is thought that conflict with another cat in the same household is the most common cause of stress in cats with F.I.C. Signs of this may not be immediately obvious and resolving or improving this can be difficult. Encouraging play and the addition of new toys to the environment can help. In cats that are indoors only, allowing some outdoor access can help (if safely possible). Consultation with a behaviourist can also be required to identify and improve causes of stress.
- Painkillers: Chronic or intermittent pain relief can be prescribed. There are various types available including non-steroidal anti-inflammatories and opiods.
- GAG replacers: GAG replacers theoretically help cats with F.I.C. by improving the abnormal bladder lining. There are multiple different formulations available and some are present in prescription diets targeted for urinary tract disease.
- Synthetic pheromone therapy: Feliway® is a feline synthetic pheromone which may help to reduce stress and anxiety. There are various ways this can be used (either a spray or a plug-in diffuser). Further advice is available from the website and there are different products available to use in different scenarios (i.e. in multi-cat homes or during times of anticipated stress).
- Anti-depressants: In extreme cases human anti-depressants can be used. These are normally reserved for severely affected cats that are refractory to all other forms of therapy.
COVID-19 UPDATE & FAQ’s
by admin on April 6th, 2020
Category: News, Tags:
Firstly may we say a massive thank you to all our customers for being so understanding and accommodating at this difficult time.
And a huge thank you to all our amazing vets, nurses and receptionists for pulling together to find the best possible means by which to provide local pets with the care they need.
Small teams are working 24/7 on a roster to provide emergency care and urgent treatment where animal welfare would be compromised by delay.
Our main Littlehampton hospital REMAINS OPEN however as you would expect we have put in place all the necessary steps to ensure that we reduce the risk of virus transmission, and are keeping the main door closed. We need to keep everyone safe.
Concerned about your pet’s health but don’t think it’s an emergency?
If you are worried about your pet, phone us and explain your situation. We’ll arrange a suitable way of conducting your appointment that is in everyone’s best interests.
There are a variety of options available to everyone – the important thing is to let us know and we’ll work together!
To respect social distancing rules, we are replacing our standard consultation service with video and telephone consultations. Please contact the practice to arrange an appointment and discuss suitable communication platforms (eg Skype, Facetime, WhatsApp). Payment can be made over the phone.
What constitutes an emergency that needs immediate care?
If any of the below apply to your pet, contact us IMMEDIATELY:
- Breathing difficulties
- Open wound injuries
- Trauma (e.g. car accidents)
- Male cats struggling in the litter tray to pass urine
- Rabbits neglecting food
- Swallowing hazards ie.toys/clothes
- Ingestion of poisonous/harmful substances
- Eye problems
- Vomiting or diarrhoea especially if your pet appears quiet or depressed or if it is going on for a long time (more than 24 hours)
- Swollen abdomen or retching (especially large dogs)
- Loss of thirst & appetite
- Struggling to give birth
- Seizuring/fitting
- Collapse
If the recommendation is to come into the practice, please follow the advice given by our staff.
- We will ask that you telephone us when you arrive in the car park.
- A member of staff will come out and take your pet into the clinic.
- After making an assessment we will ring you on your mobile to discuss the necessary treatment and way forward.
COVID-19 FAQ’s
Can I bring my pet in for an INITIAL VACCINATION COURSE?
A vaccination is not deemed an emergency and we will not be offering an initial vaccination course. If you have a puppy, we recommend that they are kept inside and not exposed to other dogs. You should book a vaccination course as soon as possible after normal service resumes.
We will review this and provide a further update if the government extends the initial three weeks of enhanced measures to slow the spread of the virus.
Can I bring my pet in for their ANNUAL VACCINATION BOOSTER?
We can postpone this vaccine for up to 3 months after the due date without requiring a restart. During these exceptional times, we ask that you delay the booster until up to a maximum of 3 months after the due date.
Can I bring in my pet for a HEALTH CHECK?
Not at this moment in time because this is not deemed critical. However, if your pet is ill we will be able to conduct a consultation via a video or telephone consultation. Please ask the practice for further information.
Can I bring my pet in for NAIL CLIPS? Or to have their ANAL GLANDS expressed?
This should be postponed if your pet is not in any danger of feeling discomfort. If you are uncertain whether this is the case, please arrange a video or telephone consultation with the vet.
How can I get my REPEAT FOOD or MEDICATIONS?
It is important that you continue to give your pet the appropriate food and medication. We are looking at ways of getting these to clients who are due them and postal / delivery services may be an option. Please contact us to make suitable arrangements.
How can I get my FLEA, WORM, TICK and FLYSTRIKE treatments?
It is important that you continue to give your pet the appropriate treatments. We are looking at ways of getting these to clients who are due them and postal / delivery services may be an option. Please contact us to make suitable arrangements.
Can my pet still have ELECTIVE OPERATIONS such as Neutering and Lumps removals?
These operations are not deemed to be critical during these extraordinary times and we therefore ask that you postpone these procedures, until government guidelines on social distancing change.
What about ULTRASOUNDS and BLOOD TESTS?
It depends whether these are deemed critical or not – please contact us to discuss.
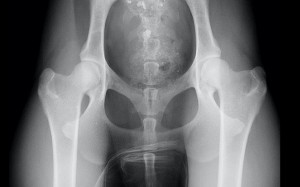
Hip dysplasia
by admin on March 3rd, 2020
Category: News, Tags:
What is hip dysplasia?
Hip dysplasia means abnormal development of the hip joint. It inevitably leads to the development of arthritis (osteoarthritis). Either the hip dysplasia or the secondary arthritis may cause hip pain.
Hip dysplasia is a genetic disorder caused by the combination of genes from the parents (dam and sire). During the first few months of life, as the hips are developing, they become unstable. As a result the ball (femoral head) and socket (acetabulum) move apart during weight bearing. This causes abnormal forces on the soft bones which leads to the ball becoming flattened and the socket becoming shallow. The process is self-perpetuating and causes damage to the covering of the bones (the articular cartilage). Cartilage damage is a key feature of the secondary osteoarthritis.
What are the signs of hip dysplasia?
Hip dysplasia is a common condition, especially in large breed dogs. The key signs are hind limb lameness and stiffness. The latter is generally most evident after rest following exercise. Difficulty rising and reluctance to jump or exercise are also common features. A rolling hind limb action may be seen in young dogs. More subtle signs include restlessness, moaning and licking the skin over the hip.
Signs tend to develop when the dog is immature and growing (five to 10 months of age) or when adult (perhaps a few years of age). When immature, it is the instability of the hip that causes the pain whereas in adult dogs it is the osteoarthritis which results in discomfort.
How is hip dysplasia diagnosed?
Examination may reveal muscle wastage (atrophy), especially over the hips. Manipulation of the joints may cause increased pain and instability may be palpable.
X-rays (radiographs) are necessary to diagnose hip dysplasia. They enable the severity of the abnormal joint development and presence of secondary osteoarthritis to be assessed.
How can hip dysplasia be treated?
The majority of dogs with hip dysplasia can be managed satisfactorily without the need for surgery. Exercise often needs to be controlled to some degree. Each dog will have its own threshold of duration and type of activity beyond which hip pain may increase. Hydrotherapy is often beneficial. Dogs that are overweight benefit from being placed on a diet. Tit-bits may need to be withdrawn and food portions reduced in size. Regular monitoring of weight may be necessary. Painkillers (anti-inflammatory drugs) may be indicated to make the dog more comfortable. Long-term drug therapy should be avoided if at all possible in view of potential side effects.
Some dogs with hip dysplasia fail to respond satisfactorily to conservative treatment and in these cases surgery may be indicated. The two key types of surgery are (1) reconstructive and (2) salvage.
Reconstructive surgery
In some young dogs (usually less than a year of age) the abnormal hip joint may be reconstructed to make it more stable. This involves cutting the pelvis and rotating the cup (acetabulum) over the ball (femoral head). The rotated section is secured in the new position with a specially designed plate. Either a triple pelvic osteotomy (TPO) or a double pelvic osteotomy (DPO) can be performed. Reconstructive surgery has the advantage of maintaining the dog’s own joint tissues and hopefully reducing the development of osteoarthritis. Unfortunately many young dogs with hip dysplasia are not good candidates for reconstructive surgery.
The aftercare following DPO or TPO surgery is very important. Exercise has to be restricted for eight weeks until the cut bones heal. Long-term hip function is generally good. Further surgery or long-term medication is generally not necessary.
Salvage surgery
Adult dogs with hip dysplasia and secondary osteoarthritis that fail to respond to medical management may require salvage hip surgery. The principle options are to replace the hip with an artificial one (total hip replacement) or to remove the ball (femoral head removal or excisional arthroplasty).
Total hip replacement generally results in significantly better limb function compared to femoral head removal and the recovery is much quicker. However, although uncommon, there are potential complications with total hip replacement surgery that need to be carefully considered prior to making a decision. With modern systems total hip replacement can be performed successfully in all sizes of patients.
Femoral head removal provides a ‘false joint’ where the limb is supported on the pelvis by scar tissue and the surrounding muscles. Recovery following surgery is slow and the limb ends up slightly shorter. Aftercare, especially physiotherapy and hydrotherapy, is very important.
Total hip replacement
Total hip replacement surgery involves replacing the painful joint with a plastic cup and a metal ball (acetabular and femoral prostheses). Care following surgery is critical to reduce the possibility of complications, such as dislocation of the prostheses. A rapid reduction in joint pain and improvement in limb function are to be expected.
What is the outlook with hip dysplasia?
The outlook or prognosis with hip dysplasia and the associated osteoarthritis is generally good. As mentioned above many dogs can be managed successfully with conservative treatment involving modification of exercise and weight, with or without the need for anti-inflammatory painkiller drugs. Those that fail to respond satisfactorily may necessitate reconstructive procedures (DPO or TPO) or salvage surgery such as total hip replacement. The outcome of these procedures is generally very good, albeit that there are potential complications.
Asthma and Bronchitis in Cats
by admin on February 3rd, 2020
Category: News, Tags:
What is asthma and feline bronchitis?
Asthma is a common respiratory condition in cats. The disease is caused by activation of inflammation in the lungs in response to irritants or allergens in the environment. The cells in the lungs respond by increased production of mucus and reversible narrowing (spasm) of the small airways. The condition is similar to the well described human form of the disease. In most cases the exact cause or trigger is unknown. A similar condition exists called chronic bronchitis. This shares many similarities with asthma. In cats with chronic bronchitis inflammation of the small airways is present however the reversible spasm is not.
What are the signs?
The signs of both conditions are similar and can vary in severity and frequency. The most common include:
- Coughing
- Rapid breathing/open-mouth breathing
- Increased noise or wheezing sounds when breathing
- Breathing difficulties/increased effort whilst breathing
Some cats can have a sudden life-threatening asthma attack. The signs of this would include breathing difficulties, open mouth breathing and weakness/collapse. This can happen in cats with previously diagnosed asthma or can be the first sign in some cases. Immediate veterinary attention is required in any cat with these signs at home.
How are the conditions diagnosed?
A diagnosis of asthma or chronic bronchitis is normally reached after a series of diagnostic tests. Often these are performed to exclude other causes of coughing/breathing difficulties in cats. Firstly a thorough history is taken. A physical examination is then performed. Harsh or wheezy sounds can be heard when the chest is auscultated.
In addition a combination of some or all of the following tests can be performed:
- Blood tests to assess general health and to evaluate white blood cell number and type.
- Faecal tests to check for feline lungworm infection may be performed.
- X-rays or a CT scan of the chest. In some asthmatic cats X-rays and/or CT scan can be normal. In others thickened airway walls are seen or signs of ‘over-inflation’ of the lungs. These tests are also important to rule out other causes of coughing/breathing abnormalities in cats.
- Bronchoscopy or video camera into the lungs to examine the inner lining of the airways.
- An airway wash may be performed. The fluid is then examined under the microscope to check inflammatory cell type and number and whether there is any evidence of bacteria or parasites. In addition the fluid is often cultured and a test for bacterial and/or parasitic DNA performed.
What is the treatment?
Both conditions are treated with anti-inflammatories (normally corticosteroids). Generally the medication is administered in one of three ways
- By injection: in cats which are hospitalised short-acting injectable forms of steroids may be administered, especially in patients that are experiencing breathing difficulties. Rarely long-acting injectable forms that can last between 3-6 weeks can be used in patients who cannot be given medication by mouth or inhaled medication.
- By mouth (tablet or occasionally liquid form): to begin with a higher dose is used before gradually reducing to the minimum effective dose.
- By inhalation: metered dose inhalers (MDIs) can be used as an alternative to oral medication. These are administered using a spacer device attached to a specially designed face mask for cats. Once a cat has become accustomed to the device it is normally well tolerated.
The advantage of this method of administration is that the steroids are delivered directly to the lungs and absorption to the rest of the body is minimised. This can reduce systemic side effects associated with oral steroid therapy. Further information regarding the Aerokat™ spacer device including a video demonstrating how it is used can be found on the following website: https://www.trudellmed.com/
In addition some cats require bronchodilators which are drugs that can help to relax (or dilate) the small airways. This medication can be given by injection in patients that are hospitalised or by mouth (tablets) at home. Inhaled bronchodilators are also available.
What is the prognosis for feline asthma?
With appropriate treatment the prognosis is normally good. Many cats will require life-long therapy. Unfortunately some cats can have progressive signs despite treatment. Uncommonly in some asthmatic cats a severe attack can be fatal.
If you have any questions about your cat’s condition, or his or her treatment, please do not hesitate to contact us.
Addison’s Disease (Hypoadrenocorticism)
by admin on January 3rd, 2020
Category: News, Tags:
What is Addison’s Disease?
Addison’s Disease is also called ‘hypoadrenocorticism’. This is a potentially life-threatening disorder caused by inadequate levels of hormones produced by small glands which are located in the abdomen (the tummy) near the kidneys. The adrenal glands produce two types of hormone that are critical for life:
- Glucocorticoids
- Mineralocorticoids
These hormones circulate through the blood stream and have effects on cells and tissues throughout the body. Dogs or cats with insufficient levels of these hormones can become very unwell.
Glucocorticoids are a natural form of cortisone (steroid). Cortisone is essential for life and must be at the right levels in the body for animals (and humans) to feel well. Steroids improve appetite and have effects on the function of the immune system that fights off infections.
Glucocorticoids can also be used as a drug for the treatment of some diseases.
Too little natural circulating cortisone is one of the components of Addison’s Disease (too much circulating glucocorticoid also causes a problem called Cushing’s Syndrome). Mineralocorticoids are also hormones produced by the adrenal glands.
Mineralocorticoids help to control the body’s ‘salt’ concentrations of both sodium and potassium. As with glucocorticoids, too much or too little mineralocorticoid in the body generally results in serious medical problems.
Addison’s Disease occurs when the body has insufficient circulating levels of both glucocorticoids and mineralocorticoids.
What causes Addison’s Disease?
Addison’s Disease results when both of the adrenal glands are damaged. This most commonly occurs when the affected animal’s own immune system, which normally fights off infections, becomes overactive and damages the adrenal glands (so called ‘immune mediated’ disease). Less common causes of Addison’s Disease are cancers or infections that can invade and kill the adrenal gland tissues.
Which animals are predisposed to Addison’s Disease?
Although Addison’s Disease is not very common, it occurs most frequently in young to middle-aged female dogs. Addison’s Disease is considered rare in cats, but the condition has been diagnosed in dogs and cats of all ages and of either sex (including neutered animals of both sexes).
Breeds that appear to be predisposed to Addison’s Disease include Portuguese Water Dogs, Standard Poodles and Bearded Collies, although it can affect any breed and crossbred dogs.
What are the symptoms (signs) of Addison’s Disease?
The signs of Addison’s Disease come on quickly, usually over a few days, although they can also appear over a period of months. Most owners notice that their pet develops several problems at about the same time including;
- Reduced appetite
- Lethargy and weakness
- Vomiting
- Diarrhoea
- Weight loss
- Tremors
In severe cases some dogs will suddenly collapse and develop shock-like symptoms.
What tests are needed to diagnose Addison’s Disease?
The signs of vomiting, diarrhoea, loss of appetite, and weight loss are extremely non-specific – many other conditions such as stomach and intestinal disease, kidney disease and pancreatic disease can cause these symptoms. Further tests are therefore needed to determine the cause of these problems.
Changes that may be noted on blood tests include changes in the salt levels in the blood – an increase in potassium and a decrease in sodium are major findings. However, these changes can also be seen with other disease processes, and if Addison’s Disease is suspected, a specific test is then recommended for confirming the diagnosis – this is called an ‘ACTH stimulation test’.
What treatment is needed if Addison’s Disease is diagnosed?
Initially, most patients with Addison’s Disease have severe dehydration and electrolyte (salt) loss, meaning that they need to be hospitalised for initial treatment and stabilisation.
Once stabilised, patients with Addison’s Disease require long term (lifelong) treatment with hormone replacement, to substitute for the missing mineralocorticoids and glucocorticoids. These drugs can be given at home in the form of tablets or injections.
The amount of medication may need to be changed over time, and frequent blood tests are recommended to monitor the condition and improve the chances of good control of the disease.
If dogs are stressed (for example due to going to boarding kennels, or because of other illness) your vet may guide you to administer some additional steroid therapy.
What’s the prognosis (outlook)?
Once dogs and cats with Addison’s Disease are correctly diagnosed and properly treated, they can live long and happy lives. Treatment is almost always successful and rewarding.
Chronic liver failure
by admin on December 2nd, 2019
Category: News, Tags:
The liver is a vital organ – animals and people cannot survive without their livers. The liver performs many complex functions that are essential to life and good health. Its main functions are:
- To convert food into useful substances such as proteins for repair and growth of the body
- To maintain normal functions such as blood clotting, fighting infection and secretion of substances to the bloodstream
- Bile production, which assists with the absorption of fat and certain vitamins
- To store the body’s primary sugar (glucose) as glycogen and be able to release it into the bloodstream when it is needed
- To neutralise and break down toxic substances such as chemicals and some medications, for later elimination in bile or through the kidneys
What is chronic liver failure?
Chronic liver failure occurs due to long-term damage to the liver, resulting in a liver that fails to work. Chronic liver failure can be caused by chronic exposure to toxins, heavy metals (copper, iron and zinc), chronic infections, chronic inflammation/irritation, cancer, blood vessel abnormalities (congenital portosystemic shunt – an abnormal blood vessel that a pet is born with that bypasses the liver), immune disease and fatty liver syndrome in cats.
The liver is very good at regenerating itself (unlike other organs such as the kidneys), but serious ongoing damage to the liver can cause long-term failure of its function – this is known as chronic liver failure. Over 75% of the liver is usually damaged before liver failure occurs.
What are the signs of chronic liver failure?
- Reduced appetite
- Lethargy
- Increased thirst
- Strange behaviours
- Vomiting
- Diarrhoea – occasionally blood in the stools, or dark, tarry stools
- Bloated tummy due to fluid accumulation
- Yellow skin, urine, gums (jaundice)
- Weight loss
How is chronic liver failure diagnosed?
Liver failure is diagnosed by blood tests which indicate dysfunction of the liver. Further investigations, such as ultrasound scans, urine cultures, blood cultures or other tests for infections, may be required in individual cases. Samples of the liver may be required to diagnose the cause of liver failure in many cases – these may be obtained through a needle placed in the liver under guidance on an ultrasound scan; alternatively the liver may be visualised by using keyhole surgery (laparoscopy) or by a full surgical procedure. The technique that is chosen will depend on a number of factors, and these will be discussed with you in the event that biopsies are required. In some cases of chronic liver disease the initial cause of the problem may no longer be present at the time of investigation, but the disease itself may still cause symptoms, and the damage to the liver may also continue.
How is chronic liver failure treated?
Chronic liver failure requires multiple treatments. It is important to try to remove the initiating cause (e.g. toxins, infections, cancer etc). Sometimes, this is not possible and it is best to support liver function and make the patient as comfortable as possible. Some diseases e.g. blood vessel abnormalities such as congenital portosystemic shunts (abnormal blood vessels that bypass the liver) can be corrected by Specialist surgical procedures.
Treatments which may be required in chronic liver failure include:
- removing fluid from the abdomen that accumulates due to scarring of the liver
- antibiotics to prevent infections
- antacids to reduce gastrointestinal ulceration
- drugs to support the liver including anti-oxidants, anti-
scarring drugs and drugs that bind to heavy metals i.e. copper and zinc - dietary modification to reduce protein and copper levels in some patients
- nutritional support – an adequate intake of vital nutrients including calories is very important in cases of chronic liver failure.
What is the long term outlook?
Considering the severity of this disease process, this is a challenging condition to treat. Early intervention and aggressive treatment can be successful in some cases where the extent of damage is not too severe. At the present time, liver transplants are not available in veterinary medicine.
If your pet develops chronic liver failure, we will discuss the treatment options in detail with you and give you the help you need in making decisions about what you wish us to do to help your pet.
Hiatal Hernia
by admin on November 5th, 2019
Category: News, Tags:
What is a Hiatal Hernia?
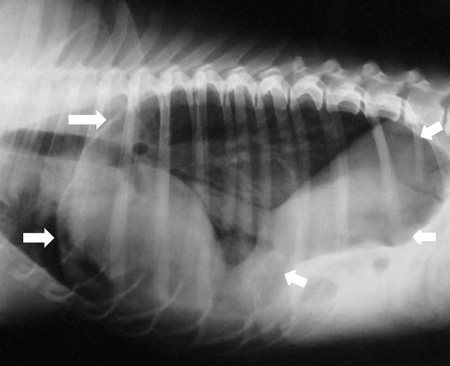
A hiatal hernia is the abnormal movement of part of the stomach from its normal position in the abdomen into the chest.
The diaphragm is a sheet of muscle which separates the chest and abdomen. The oesophagus (food pipe) and major blood vessels and nerves pass through the diaphragm via small openings within the muscle. During the development of a puppy, these openings may be excessively large, allowing organs which should remain in the abdomen to pass into the chest. The stomach can become permanently displaced but more commonly slides back and forth between the abdomen and the chest.
What breeds are affected?
English Bulldogs are the breed most commonly seen with this condition in the UK. Brachycephalic dogs (those with a shortened nose such as English Bulldogs, French Bulldogs and Pugs) are at an increased risk of displacement of the stomach into the chest as the negative pressures within the chest created by their abnormal airway, act to ‘suck’ the stomach through the opening in the diaphragm.
What are the symptoms?
Dogs with hiatal hernia frequently vomit or regurgitate (passive reflux of stomach contents) due to the abnormal position of the stomach. These signs most commonly occur during peak exercise or when the dog becomes excited. Reflux of acidic stomach contents into the oesophagus results in inflammation (reflux oesophagitis) which can trigger a self-perpetuating cycle of regurgitation causing further inflammation.
Other symptoms which may be observed include excessive salivation, poor appetite and weight loss. Vomiting and/or regurgitation of stomach contents can result in inhalation of this material and the development of secondary pneumonia. In this scenario symptoms such as a cough, nasal discharge and breathing difficulties will be observed.
How is the diagnosis made?
Hiatal hernia is most commonly diagnosed via fluoroscopy (a moving x-ray study). Whilst the dog is eating, a continuous run of X-rays are taken which allows the swallowing process and position of the stomach to be closely observed. Typical findings consistent with a diagnosis of hiatal hernia are food entering the stomach and then passing back up the oesophagus (reflux) along with movement of the stomach back and forth across the diaphragm.
As the stomach typically slides back and forth from a normal to abnormal position, there is the possibility that the imaging study documents a normal stomach position. This does not exclude the diagnosis of hiatal hernia as the most likely explanation for a normal stomach position is that the stomach has not been ‘captured’ in an abnormal position at the time of the imaging study. If this scenario arises, a presumptive diagnosis of hiatal hernia may be made based on the breed of the dog and the symptoms.
What treatment options are available?
The management of hiatal hernia can be broadly divided into medical and surgical options.
Medical management consists of the use of medication to reduce inflammation within the oesophagus (antacids) and aid motility of the gastrointestinal tract. Medication often reduces the severity and frequency of the symptoms but is unlikely to eliminate them completely due to the persistently abnormal position of the stomach.
Surgical correction is often indicated to resolve the clinical problems. In some patients surgical correction of the upper airway problems, as seen in Brachycephalic dogs, eliminates the problems. In other patients the surgery will consist of reducing the size of the overly large opening in the diaphragm and stitching the oesophagus and stomach permanently in a normal position. This technique prevents movement of the stomach into the chest.
What is the outlook (prognosis) for my dog?
The prognosis following surgery is good. The aim is to eliminate the symptoms (regurgitation and vomiting) completely. Rarely, dogs may continue to regurgitate and/or vomit after surgery although with a markedly reduced frequency compared to before surgery. If this is the case, these mild symptoms can usually be controlled with the use of medication.